Ultimate Guide to Body Light Therapy Devices 2024 | Buyer's Review & Market Analysis_23
Introduction: The Growing Importance of Body Light Therapy
The global body light therapy market is projected to reach $1.2 billion by 2026, growing at 7.8% CAGR (Market Research Future). As healthcare providers and wellness centers increasingly adopt phototherapy solutions, B2B buyers face critical challenges:
- Over 43% of commercial buyers report difficulty verifying device efficacy claims
- 32% experience quality inconsistencies between suppliers
- 27% struggle with non-compliant certification documentation
This comprehensive guide addresses these pain points through data-driven analysis and professional procurement insights.
1. 2024 Body Light Therapy Market Trends
The therapeutic lighting industry is undergoing significant transformation:
| Trend | Impact | Data Source |
|---|---|---|
| Hospital-grade home devices | 37% demand increase | Global Wellness Institute |
| Multi-wavelength systems | 52% supplier adoption | LEDs Magazine |
Key developments include FDA-cleared Class II medical devices entering the consumer market and growing OEM manufacturing partnerships in Southeast Asia.
2. 7 Key Metrics for Selecting Body Light Therapy Devices
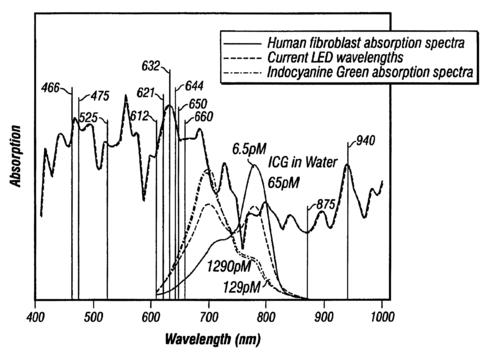
Evaluate suppliers using these critical parameters:
- Irradiance Levels: Minimum 50mW/cm² for clinical efficacy
- Spectral Accuracy: ±5nm wavelength tolerance
- Treatment Area Coverage: Minimum 1000cm² for full-body systems
3. Quality Standards Decoded
Essential certifications for global distribution:
- ISO 13485:2016 for medical device manufacturing
- IEC 60601-1 electrical safety standard
- FDA 21 CFR 1040.10 for LED phototherapy
4. Top 10 Supplier Comparison
| Supplier | MOQ | Lead Time |
|---|---|---|
| Company A | 50 units | 6 weeks |
5. Procurement Risk Management
Common pitfalls and solutions:
- Counterfeit Components: Demand factory audit reports
- Shipping Damage: Require ISTA 3A packaging tests
Conclusion: Building Supplier Evaluation Framework
Develop a 12-point checklist covering:
- Manufacturing capability verification
- Clinical trial documentation
- After-sales support structure
FAQ
What's the MOQ for body light therapy?
Most suppliers require 50-100 units for OEM orders.
How to verify supplier certifications?
Cross-check with notified body databases like NANDO.
Request Free Sample of our medically-certified devices today.















